In absorption (also known as gas scrubbing, gas absorption, or gas washing), one or more species transfer from the gas phase to a liquid solvent. Solutes or absorbates are the species that are transferred to the liquid phase. In order to separate gas mixtures, remove contaminants, or recover valuable substances, absorption is utilized. In the stripping process, absorbed solute from the solvent is extracted. Absorbers are typically employed in conjunction with strippers to enable recovery (or regeneration) and recycling of the absorbent. When utilized as the absorbent, water is often isolated from the solute by distillation rather than stripping.
The absorption process can be performed for any of the following purposes:
a) Separation of high-value components
b) As a step in the synthesis of certain chemicals
c) To get rid of undesirable elements (pollutants)
1 Types of Absorption
1.1 Physical Absorption
Physical equilibria control physical absorption, and the mass transfer proceeds only by diffusion.
1.2 Chemical Absorption
In this absorption type, a chemical reaction starts as soon as a certain element comes into touch with the liquid that is absorbing it. The diffusion rate is then improved by lowering the component proportion in the liquid phase.
2 Types of Absorber Column
For purposes of absorption, there are two basic categories of absorbers: Plate Column and Packed Column.
2.1 Plate Column
The plate column has the following characteristics:
a) Various gas and liquid flow rates can be handled using plate columns.
b)More precise prediction of plate efficiency.
c) The plate column operates very smoothly.
d) The plate column can have a cooling arrangement.
e) The plate column provides stage-wise contact.
f) Even if the tray is sealed off, it is simpler to clean when the liquid produces fouling or the deposition of particulates, and manholes can be installed above the plate.
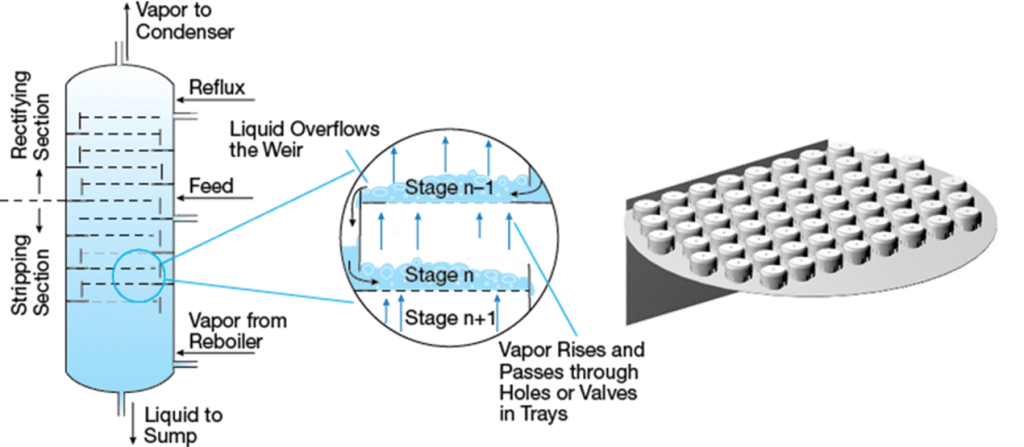
Figure 1: Plate column schematic
2.2 Packed Column
The characteristics of the packed column are given below:
a) Low-capacity activities are better suited for packed columns.
b) The field of vacuum distillation benefits greatly from packed columns. Column pressure loss is crucial to reduce the temperature and pressure at the bottom of the column.
c) Packed columns facilitate low liquid hold-up, making them suitable for separating compounds sensitive to heat.
d) Packing can be the sole solution if corrosion is an issue.
e) A packed column has a lower pressure drop per unit length.
f) The packed column keeps the liquid and vapor phases in constant contact.
g) Packed columns can handle the foaming system.
h) Almost every material is a viable option for modern high-capacity pickings.
i) The employment of low-weight and high-capacity packing results in a packed column with a lower overall weight.

Figure 2: Packed column schematic
2.3 Disadvantages of Plate and Packed Absorber Columms
2.3.1 Plate Column
a) Plate columns operate worse when using a foaming system. Anti-foaming substances can be used to reduce foam.
b) The liquid holds up quite well. Consequently, there is a significant pressure reduction.
c) The price of the plate column for corrosive liquids is costly owing to the usage of corrosion-resistant material.
d) The required supporting structure is relatively costly.
2.3.2 Packed Column
a) Predictions of HTU (separation effectiveness of the particular packing) and HETP (packing height that can provide same separation as an equilibrium stage) could not be particularly accurate.
b) For very low liquid flows, packed columns are not appropriate.
c) It is challenging to provide cooling arrangements in densely packed columns.
d) In a filled column, side streams cannot be removed.
3 The Principal Requirements of Packing
a) It must be chemically inert to the fluids intended to use in the tower.
b) It has to be strong but not too heavy.
c) It must have sufficient channels for both streams that do not cause excessive liquid hold-up or pressure loss.
d) It must ensure that the liquid and gas make excellent contact.
e) The price has to be reasonable.
Most packaging comprises harmless, low-cost, and relatively light materials like clay, graphite or porcelain. Some limes employ thin-walled metal rings consisting of steel or aluminum. Some common packings are shown in Figure 3.

Figure 3: Some common packing types
4 Important Steps in Absorption Column Design
An absorber column design involves several processes requiring theoretical and practical expertise. Some selection criteria or designing steps have been elaborated in the above sections (e.g., column type selection, packing types, etc.), and some steps are added here with brief details. Steps that require complex calculations are skipped here to enhance understandability.
a) Determining the estimated column diameter
b) Selecting the column type
c) Determining the material and packaging
d) Estimating the packing size
The largest size of packing that is frequently utilized is 50mm. Costs for smaller sizes are noticeably more than for bigger sizes. Poor liquid distribution and decreased absorption rate might result from too much packing in a small column. Size ranges that are recommended in the literature are:

e) Calculating column actual diameter
f) Calculating the flooding velocity
Depending on a column’s loading point, flooding velocity is determined. It is when the gas velocity is high enough to cease the flow of the liquid. Once the flooding point is reached, the pressure begins to fall considerably more quickly when the gas absorbs all the liquid. The flooding velocity is the gas velocity at this location (limiting velocity).
McCabe & Smith stated that the operating velocity should be between 50% to 90% of the flooding velocity.
g) Determining the no. of transfer units (NoG)
The Cornell Method and the Onda Method are the two approaches that can be applied. The following illustration shows the steps:

Compared to Onda’s method, Cornell’s is much simpler because some of the values have been presumptive. Though it may take longer to compute, Onda’s Method offers a more precise design.
h) Measuring the packing’s height
i) Determining the column’s height
j) Measuring pressure decrease
Each step that has been described here contributes significantly toward the final column design.
5 References
- Laso, M., & von Stockar, U. (2000). Absorption. Kirk‐Othmer Encyclopedia of Chemical Technology.
- Foust, A. S., Wenzel, L. A., Clump, C. W., Maus, L., & Andersen, L. B. (2008). Principles of unit operations. John Wiley & Sons.
- Noble, R., & Terry, P. (2004). Adsorption. In Principles of Chemical Separations with Environmental Applications (Cambridge Series in Chemical Engineering, pp. 182-213). Cambridge: Cambridge University Press.

 To all knowledge
To all knowledge